IMPORTANT SAFETY INFORMATION
Contraindications
Corlanor® is contraindicated in patients with acute decompensated heart failure, clinically significant hypotension, sick sinus syndrome, sinoatrial block, 3rd degree atrioventricular block (unless a functioning demand pacemaker is present), clinically significant bradycardia, severe hepatic impairment, pacemaker dependence (heart rate maintained exclusively by the pacemaker), and concomitant use of strong cytochrome P450 3A4 (CYP3A4) inhibitors.
Fetal Toxicity
Corlanor® may cause fetal toxicity when administered to a pregnant woman based on embryo-fetal toxicity and cardiac teratogenic effects observed in animal studies. Advise females of reproductive potential to use effective contraception when taking Corlanor®.
Atrial Fibrillation
Corlanor® increases the risk of atrial fibrillation. The rate of atrial fibrillation in patients treated with Corlanor® compared to placebo was 5% vs. 3.9% per patient-year, respectively. Regularly monitor cardiac rhythm. Discontinue Corlanor® if atrial fibrillation develops.
Bradycardia and Conduction Disturbances
Adult Heart Failure Patients
Bradycardia, sinus arrest and heart block have occurred with Corlanor®. The rate of bradycardia in patients treated with Corlanor® compared to placebo was 6% (2.7% symptomatic; 3.4% asymptomatic) vs. 1.3% per patient-year, respectively. Risk factors for bradycardia include sinus node dysfunction, conduction defects, ventricular dyssynchrony, and use of other negative chronotropes. Bradycardia may increase the risk of QT prolongation which may lead to severe ventricular arrhythmias, including torsades de pointes, especially in patients with risk factors such as use of QTc prolonging drugs.
Concurrent use of verapamil or diltiazem also increases Corlanor® exposure, contributes to heart rate lowering, and should be avoided. Avoid use of Corlanor® in patients with 2nd degree atrioventricular block unless a functioning demand pacemaker is present.
Pediatric Heart Failure Patients
Bradycardia and first-degree heart block were observed in pediatric patients treated with Corlanor®. Asymptomatic and symptomatic bradycardia were observed in 6.8% and 4.1% of pediatric patients treated with Corlanor®, respectively. In the placebo treatment arm, 2.4% of pediatric patients had asymptomatic bradycardia, but none had symptomatic bradycardia. Bradycardia was managed through dose titration but did not result in study drug discontinuation.
Adverse Reactions
Adult Heart Failure Patients
The most common adverse drug reactions reported at least 1% more frequently with Corlanor® than placebo and that occurred in more than 1% of patients treated with Corlanor® were bradycardia (10% vs. 2.2%), hypertension or increased blood pressure (8.9% vs. 7.8%), atrial fibrillation (8.3% vs. 6.6%), and luminous phenomena (phosphenes) or visual brightness (2.8% vs. 0.5%).
In postmarketing experience, torsades de pointes has been observed.
Pediatric Heart Failure Patients
Bradycardia (symptomatic and asymptomatic) occurred at rates similar to those in adults. Phosphenes were observed in pediatric patients treated with Corlanor®.
Please see full Prescribing Information and Medication Guide.
INDICATIONS
Heart Failure in Adult Patients
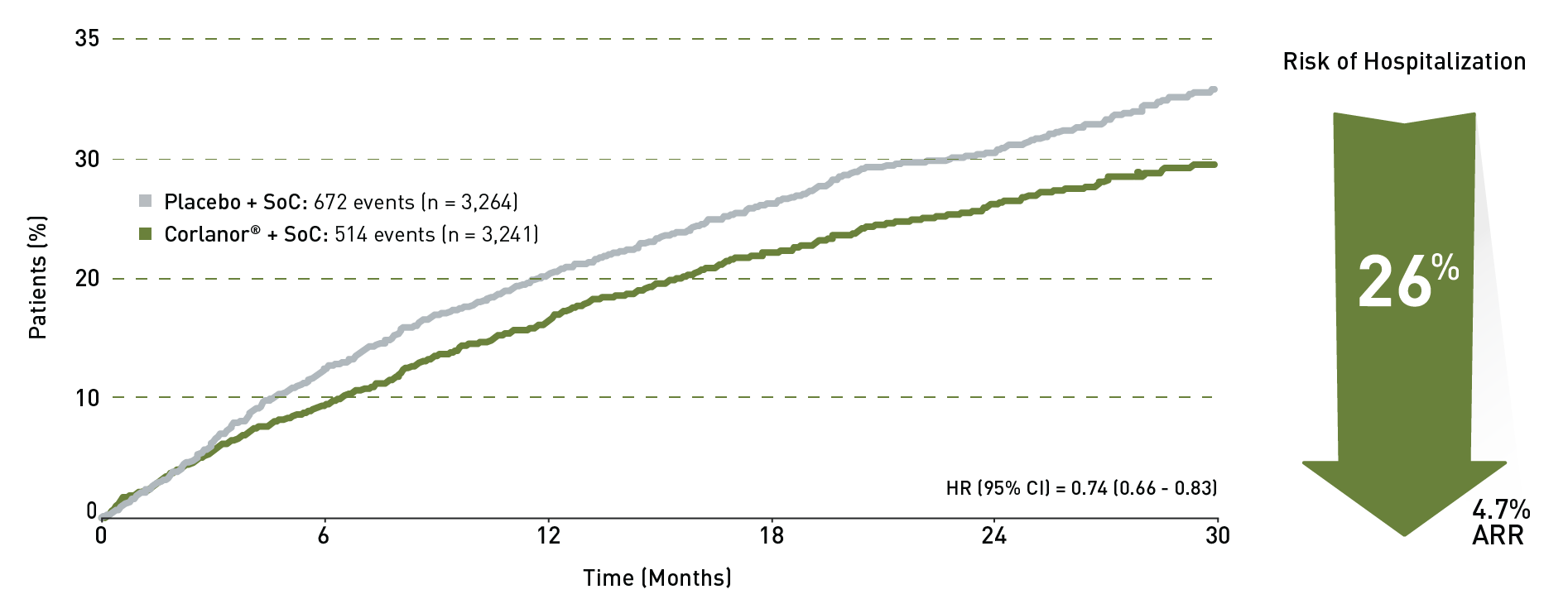
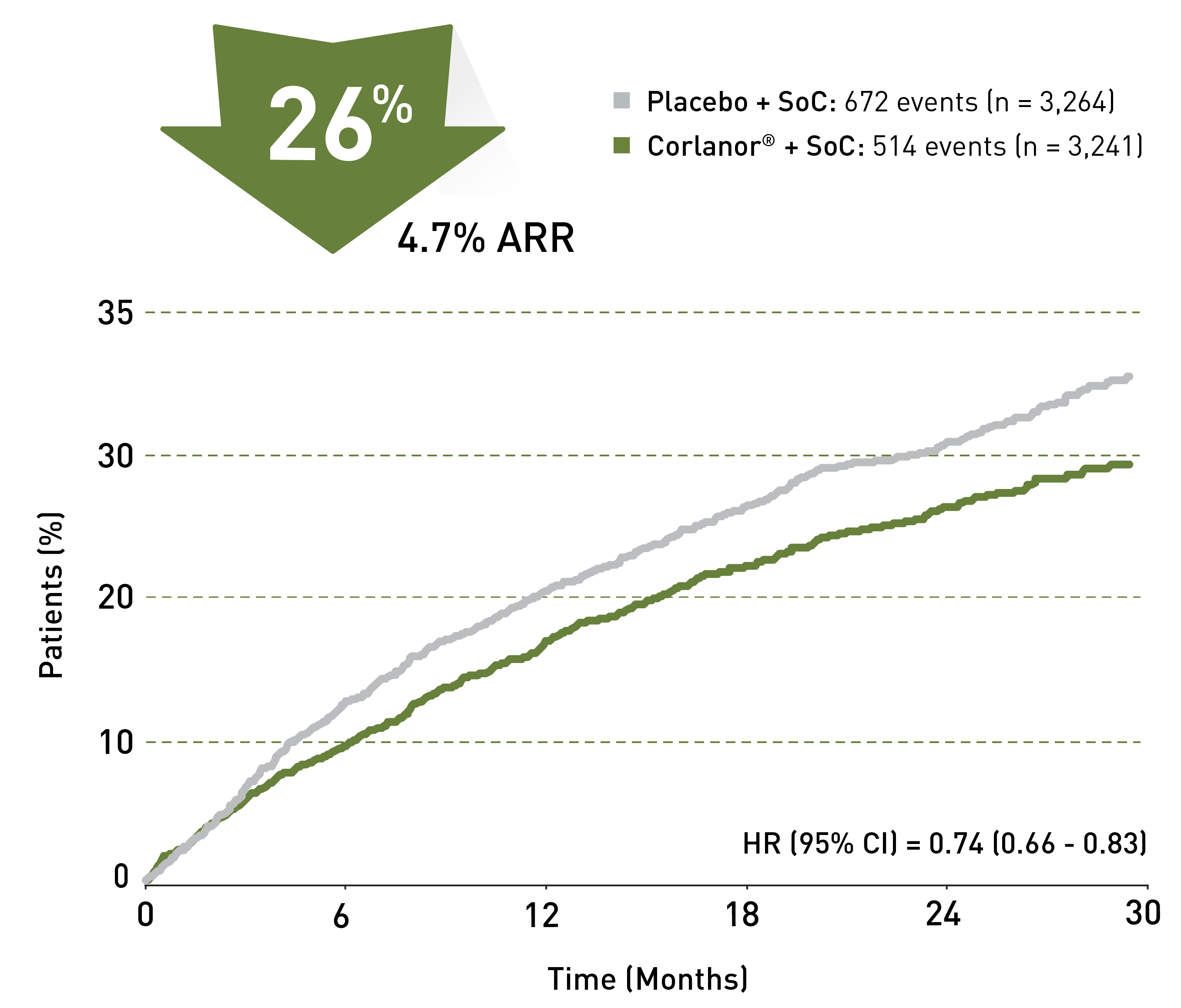
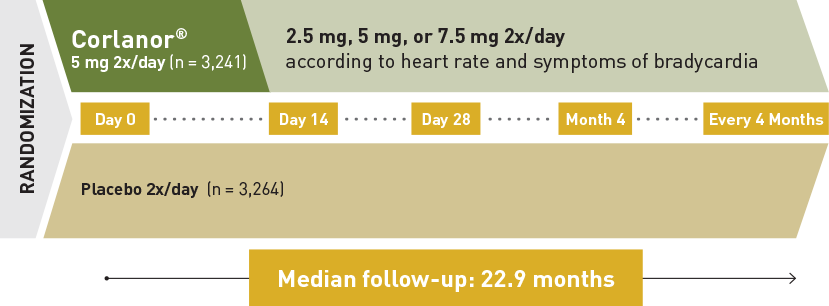
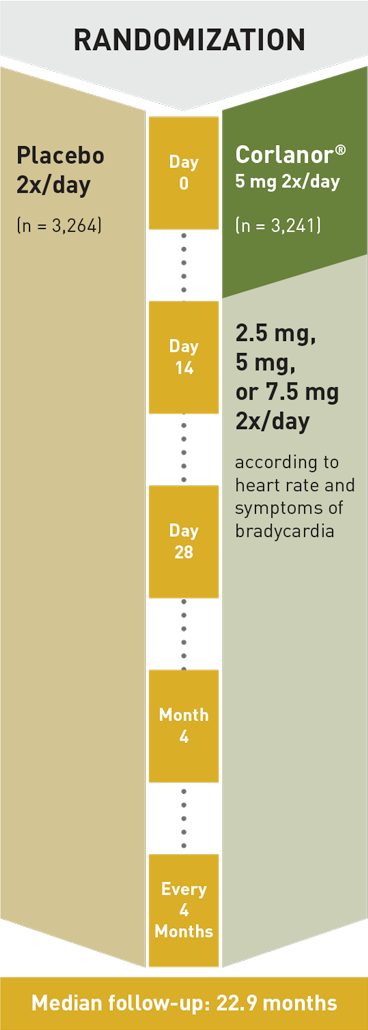
Corlanor® is indicated to reduce the risk of hospitalization for worsening heart failure in adult patients with stable, symptomatic chronic heart failure with left ventricular ejection fraction ≤ 35%, who are in sinus rhythm with resting heart rate ≥ 70 beats per minute and either are on maximally tolerated doses of beta-blockers or have a contraindication to beta-blocker use.
Heart Failure in Pediatric Patients
Corlanor® is indicated for the treatment of stable symptomatic heart failure due to dilated cardiomyopathy (DCM) in pediatric patients aged 6 months and older, who are in sinus rhythm with an elevated heart rate.